Topics: Biology, Biotechnology, DNA, Molecules
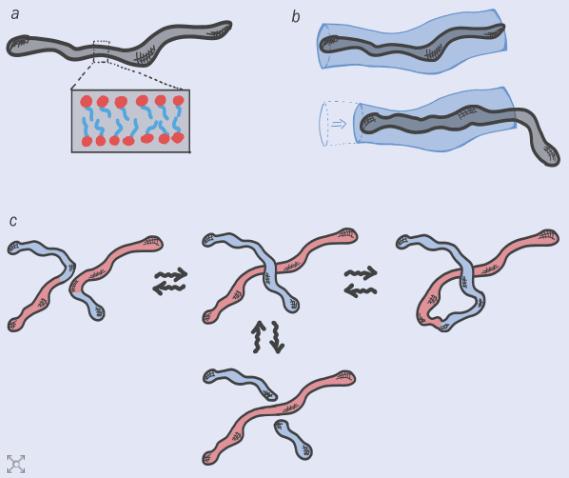
DNA molecules are not fixed objects – they are constantly getting broken up and glued back together to adopt new shapes. Davide Michieletto explains how this process can be harnessed to create a new generation of “topologically active” materials.
Call me naive, but until a few years ago I had never realized you can actually buy DNA. As a physicist, I’d been familiar with DNA as the “molecule of life” – something that carries genetic information and allows complex organisms, such as you and me, to be created. But I was surprised to find that biotech firms purify DNA from viruses and will ship concentrated solutions in the post. In fact, you can just go online and order DNA, which is exactly what I did. Only there was another surprise in store.
When the DNA solution arrived at my lab in Edinburgh, it came in a tube with about half a milligram of DNA per centimeter cube of water. Keen to experiment with it, I tried to pipette some of the solutions out, but they didn’t run freely into my plastic tube. Instead, it was all gloopy and resisted the suction of my pipette. I rushed over to a colleague in my lab, eagerly announcing my amazing “discovery”. They just looked at me like I was an idiot. Of course, solutions of DNA are gloopy.
I should have known better. It’s easy to idealize DNA as some kind of magic material, but it’s essentially just a long-chain double-helical polymer consisting of four different types of monomers – the nucleotides A, T, C, and G, which stack together into base pairs. And like all polymers at high concentrations, the DNA chains can get entangled. In fact, they get so tied up that a single human cell can have up to 2 m of DNA crammed into an object just 10 μm in size. Scaled up, it’s like storing 20 km of hair-thin wire in a box no bigger than your mobile phone.
Make or break: building soft materials with DNA, Davide Michieletto is a Royal Society university research fellow in the School of Physics and Astronomy, University of Edinburgh, UK